The Hallmarks of Aging – Key Mechanisms and Models in Neurobiology
Introduction: The History of “Hallmarks” in Biological Research
Life science researchers worldwide are familiar with the publication series “The Hallmarks of Cancer”, initially laid out by Hanahan and Weinberg (2000) and expanded over subsequent decades to map key biological capabilities acquired during the development of human cancer. This organizing framework has provided an excellent view of an immensely complex disease progression. Fortunately, several other well-considered compilations of life science research area hallmarks have since been published, each offering a cohesive way to approach a diverse range of molecular drivers.
In this article, we are inspired by “The Hallmarks of Aging” (López-Otín et al., Cell, 2013) and its 2023 update “Hallmarks of Aging: An Expanding Universe” (López-Otín et al., Cell, 2023), exploring cellular processes that drive aging — from genomic instability and epigenetic alterations to inflammation and stem cell exhaustion. Given that aging is a major “risk factor” for cancer, cardiovascular disease, and neurodegenerative disorders, the more we learn about the biology of aging, the greater the potential for wide-ranging insights applicable to human health and longevity.
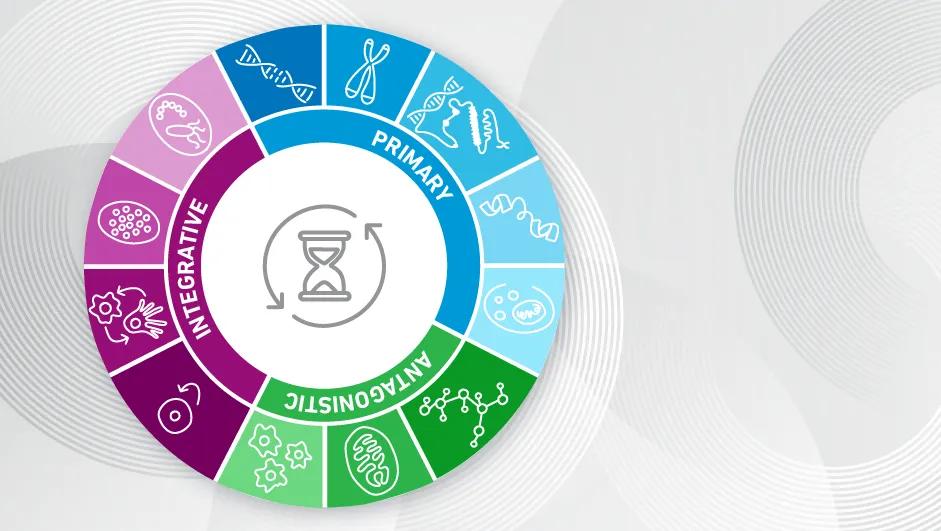
The current list of hallmarks of aging is robust, with 12 to date, however, López-Otín and colleagues have identified commonalities that allow their organization into three categories:
Primary Hallmarks: Events that cause damage to cells – be it to the genome, the chromosomes, epigenome, proteome or other cellular components, all contributing to their loss of integrity and development of age-related disease.
Antagonistic Hallmarks: Those mechanisms that reflect the cell response(s) to damage from the primary hallmarks, typically acting to keep damage in check, but which may become overly active or chronic enough to drive accumulation of senescent cells.
Integrative Hallmarks: A phase and phenotype that occurs when cells can no longer effectively counter the challenges posed by primary or antagonistic events, leading to states of long-term exhaustion, chronic inflammation, imbalance, or dysbiosis.
In the sections that follow, we will walk through notable features of the primary aging hallmarks and spotlight JAX mouse models valuable for investigating these processes in depth, with a focus on the close relationship between aging and the development of neurodegenerative disease.
Primary Hallmarks of Aging
As the name implies, factors that cause damage within cells are a primary path to their demise. Cells have processes in place to counter this, but as healthy cells age, they become more susceptible to damage – whether through loss of repair efficiency, accumulation of damage over time to an unsustainable level, or through epigenetic modifications – the mapping of which have led to some tantalizing “epigenetic clocks” to measure biological age (Kabacik et al., 2022). Some mechanistic ways this damage manifests in cells include changes in the structure and integrity of DNA and chromatin, shortening of the protective telomeric ends of chromosomes, or buildup of damaged proteins — to name a few. Let's dive into some key examples of primary hallmarks of aging:
Genomic Instability: Our cells are constantly under attack from forces that threaten their DNA. Some of these stresses come from outside, including insults like chemical exposure, UV irradiation, or physical damage, while others arise from within the cell itself, such as errors made during DNA replication. When cells are young and healthy, they are well equipped to handle these challenges, using efficient DNA repair systems to keep damage under control. But as time goes on, those protective mechanisms begin to wear down. Damage starts to build up in the nucleus and mitochondria, and in some cases, DNA even appears where it doesn't belong, floating in the cytosol. This accumulation of damage is a key driver of cellular aging. In fact, genomic instability isn't just a hallmark of aging; it's also a defining feature of cancer, underscoring why aging is widely regarded as a major risk factor for the disease.
Model Spotlight: JAX Ercc1- mice carry a targeted knockout of the murine DNA repair gene Ercc1 (excision repair cross-complementing group 1). This model displays features of accelerated aging in multiple tissues, including liver, kidney, and the central nervous system, making it well suited for research into aging and neurobiology.
Telomere Attrition: Chromosomes bear protective caps at their ends, namely regions of nucleotide sequence repeats and associated proteins that make up telomeres. These regions help maintain chromosome integrity as DNA replication machinery fails to function at the very tips of the chromosome. Every time a cell divides, it loses a bit of telomere length, so that telomeres essentially provide a small buffer that can be sacrificed with each round of mitosis. In germ cells, specialized telomerase enzymes (TERT, TERC) act protectively by adding new telomeric DNA to chromosomal ends. But telomerase function diminishes in mature somatic cells, so that with repeated cell division over time, telomeres shorten enough to trigger cell senescence. Interestingly, cancer cells can reactivate telomerase enzymes, rebuilding their protective telomere ends as one of the many ways they evade normal cellular mechanisms to become immortal.
Model Spotlight: JAX TERT and TERC polymerase knockout models are powerful tools for studying aging and cancer mechanisms, as demonstrated in a recent publication by Akincilar et al. (2025), which incorporates both models for analysis of telomerase activity. Another recent publication by Liu et al. (2026) leverages the TERC KO model to demonstrate the effect of telomere shortening in triggering microglial senescence and neuronal dysfunction.
Learn more from JAX’s “Minute to Understanding: What are Telomeres?"
Epigenetic Alterations: Epigenetics is the study of chemical modifications to DNA, histones and chromatin – changes that regulate if and when genes are turned on. The basic mechanisms of epigenetics involve a system of regulated modifications and the enzymes that recognize, add, or remove them – the "readers, writers, and erasers." The most common epigenetic changes researchers track and map to gene on/off status include DNA methylation and the methylation/acetylation of histone proteins that form the core of nucleosomes (DNA wrapped around histones to provide packaging and regulation of DNA accessibility). Age-related changes in epigenetic modifications have been observed for decades, and numerous "epigenetic clocks" have been developed and published, each proposing the ability to predict biological age based on patterns of DNA methylation and other factors. Of particular interest in the field of histone modifications that impact aging are sirtuin (SIRT) proteins, which function as histone deacetylases (HDACs). SIRTs are key factors in aging and lifespan research because of their demonstrated role in the delay of cellular senescence (Simon et al., 2022).
Model Spotlight: Sirt1loxP has been leveraged for age-induced neurodegenerative disease modeling of ALS by Herskovitz et al., 2018 and by the laboratory of David Sinclair for analysis of NAD+-H2S signaling as a cause of vascular aging (Das et al., 2019).
JAX models are available for a range of sirtuin gene-targeted knockouts and strains engineered to support sirtuin conditional KO research.
Loss of Proteostasis: The proteome represents a cell's normal, requisite suite of proteins expressed and maintained for cell function and homeostasis. As cells age or experience damage as described above, an imbalance in the level and composition of the cell protein inventory can occur. Disruption in how proteins are transcribed or processed can lead to improper folding, loss of timely degradation and disposal, or inappropriate accumulation that overwhelms normal cellular clearance mechanisms. Neurodegenerative disorders are important examples of age-related disease states that result from damaged or incorrectly folded proteins accumulating to form toxic plaques, as is characteristic of ALS, Alzheimer's, and Parkinson's disease. The close tie between hallmarks of aging and the hallmarks of neurodegenerative disease are evident with this core hallmark.
Model Spotlight: The Apolipoprotein E (ApoE) protein is critical for transporting lipids in blood and in the brain, where it can be produced locally by astrocytes. APOE4, a variant of ApoE, is one of the most prominent genetic risk factors for Alzheimer’s disease, as it is less efficient at clearing beta-amyloid proteins, allowing their accumulation into plaques that disrupt brain function. JAX mouse models have been used to develop an APOE4 knock-in platform and proposed preclinical model for dysregulated proteostasis in Alzheimer’s disease (Shin et al., 2025).
Learn about JAX’s role in the MODEL-AD Consortium for Alzheimer’s disease research.
Disabled Macroautophagy: Autophagy is the cell’s ability to “eat” itself (or part of itself)! It is a well-orchestrated process in which autophagosomes and other vesicles act as membrane-bound pockets, moving through the cell and fusing together to collect damaged targets and deliver them to digestive enzymes for breakdown and removal. This intracellular recycling system is critical to maintaining a healthy cytosol and plays a key role in removing damaged cellular components, including damaged and misfolded proteins described above in the context of proteostasis. This recycling system serves not only to break down and remove macromolecules, but also to reclaim their components for reuse. Autophagosomes and lysosomes work together to digest items as large as organelles into amino acids and nucleotides that can be re-purposed. Autophagy is also used by cells to destroy pathogens or to expel cell waste via fusion with the cell membrane. What happens when this key functionality winds down? In aging cells, we see exactly that – an accumulation of toxic, overproduced, and under-cleared macromolecules that can trigger disease pathology.
Model Spotlight: In recent work by Jia et al. 2026, the authors demonstrate that clearance of Tau proteins is reduced in tauopathy neurons. The mechanism they present involves a novel form of contact and communication between the mitochondria and autophagic vesicles; in healthy cells, these vesicles normally clear Tau proteins, but in tauopathy neurons, they instead become “tethered” and less able to move and function. JAX Tau mouse models were instrumental to this work.
Integrative and Antagonistic Hallmarks of Aging
While it is beyond the scope of this article to review the suite of hallmarks in the Integrative and Antagonistic categories, we have called out a key hallmark from each group that is particularly interesting in the context of neurodegenerative disease research.
Featured Integrative Hallmark – Dysbiosis: We hear so much about a healthy microbiome — the community of microorganisms that populate our skin, nasal and oral cavities, and the gut. While often thought of in relation to personal nutrition and a healthy lifestyle, the composition of our microbiome and its disruption are now recognized as having a significant impact on the development of disease – from cardiovascular disease (the “gut-heart axis”) to neurodegenerative disease. Dysbiosis is the imbalance of a normal and healthy microbiome and can be triggered by a range of external factors: diet, exercise, medications...you name it! It is also true that our microbiome evolves and becomes less diverse as a function of aging, so that the pathology associated with dysbiosis soon follows, so much so that this phenomenon is recognized as a primary hallmark of aging.
Model Spotlight: JAX Model 5XFAD, which overexpresses mutant human amyloid beta (A4) precursor protein 695 (APP), with familial Alzheimer's Disease (FAD) mutations, was leveraged by Yang et al., 2026 in a compelling study on the connection between dysbiosis, aging, and Alzheimer’s disease. Their work shows that gut microbiota from aged mice is transferable to young mice via co-housing, leading to gut dysbiosis and triggering amyloid pathology in the young cohort. Interestingly, probiotic intervention was shown to reduce these effects, highlighting the importance of the microbiome and dysbiosis in aging and AD pathology, as well as offering some potential approaches for reversing the pathology.
Learn about the microbiome in aging and “the fountain of youth bacteria” in our presentation "Gut Check: Is the Microbiome the Key to Your Health?"
Featured Antagonistic Hallmark – Stem Cell Exhaustion: We have reviewed how aging affects normal somatic cells, leading to a decline in beneficial cellular processes as well as the deleterious overactivation of repair processes. Stem cells are similarly impacted, progressively losing their regenerative and self‑renewal capacity. This decline impacts nearly every tissue and organ in the body and becomes especially noticeable in highly regenerative tissues, such as hair follicles, which contribute to hair loss, and epidermal cells, which underlie the very unfavorable signs of aging skin. Many of these changes are driven by core hallmarks of aging, including accumulated DNA damage, telomere shortening, and altered gene regulation, all of which progressively reduce stem cell plasticity and regenerative potential. In the brain, aging neural stem cells (NSCs) lose their ability to mature into fully functional neurons, contributing directly to the progressive neurodegeneration characteristics of Alzheimer’s and Parkinson’s disease.
Cell Model Spotlight: In partnership with the iPSC Neurodegenerative Disease Initiative, a project from the Center for Alzheimer's and Related Dementias (NIH), and with support from the Chan Zuckerberg Initiative, and Aligning Science Across Parkinson's (ASAP), The Jackson Laboratory now offers a catalog of human induced pluripotent stem cells (iPSCs) for Alzheimer's and related Dementias (ADRDs), ALS, and other neurodegenerative disorders.
Our range of mouse models for neuroscience research comprises highly published, innovative platforms to study Alzheimer’s, Parkinson’s, and tauopathy diseases. Learn more about these models here. JAX has also developed study-ready aged C57BL/6J (B6) mice to support a range of age-related research areas, including longevity and cancer.
Resources for Aging & Neurodegenerative Disease Research
JAX Center for Aging Research: https://www.jax.org/research-and-faculty/research-centers/aging-center
JAX Blog: Five Must-Reads If You Study Aging in Mice
JAX Webinar: Research Using Aged B6 Mice – Considerations, Applications, and Best Practices