Immune Cell Humanized Mice: CD34+ Hematopoietic Stem Cell Engrafted Mice
Charu Gupta, Ph.D., Senior Technical Information Scientist
Key Takeaways:
Diverse and Functional Immune Populations: HSC engraftment supports maturation of both adaptive and innate immune populations for a more complete in vivo human immune system.
Long Experimental Window: HSC engraftment supports long-term longitudinal in vivo investigations (e.g., immune memory formation, rechallenge studies, gene and cell therapy evaluation, and co-engraftment of human tumors).
NSG™ variants: Several NSG variants are available, each engineered to express specific human cytokines and/or growth factors that support different immune populations at defined relative frequencies. This enables tailored immune profiles suited to different experimental applications.
Immune cell humanized mice are a valuable model for investigating human immune biology, disease progression, and therapeutic responses in vivo. These models are genetically engineered to lack a functional mouse immune system (i.e., immunodeficient) and express human cytokines and growth factors that support the survival, maturation, and differentiation of engrafted human cells into functional leukocyte populations. Immune humanized mice help bridge the translational gap by recapitulating key aspects of human physiology and pathology within a biological complex system.
At JAX, we specialize in two well-validated methodologies for generating immune cell humanized mice, both of which reliably recapitulate many aspects of the human immune system in a reproducible manner. These approaches are:
JAX performs both types of engraftment in multiple genetic variants of NSG™ mice and has optimized both approaches to efficiently and consistently generate humanized mice. While both engraftment approaches result in immune cell humanized mice, there are distinct differences between the two models that influence the design and outcome of experiments. Understanding the differences between these two approaches will help you choose the optimal immune humanized model for your research.
In this blog, we’ll explore what HSCs are and the advantages they offer for your research. For a more detailed look into PBMC engraftment and its experimental uses, check out our related PBMC engraftment blog. These two blogs aim to help you select the most suitable engraftment method for your research needs.
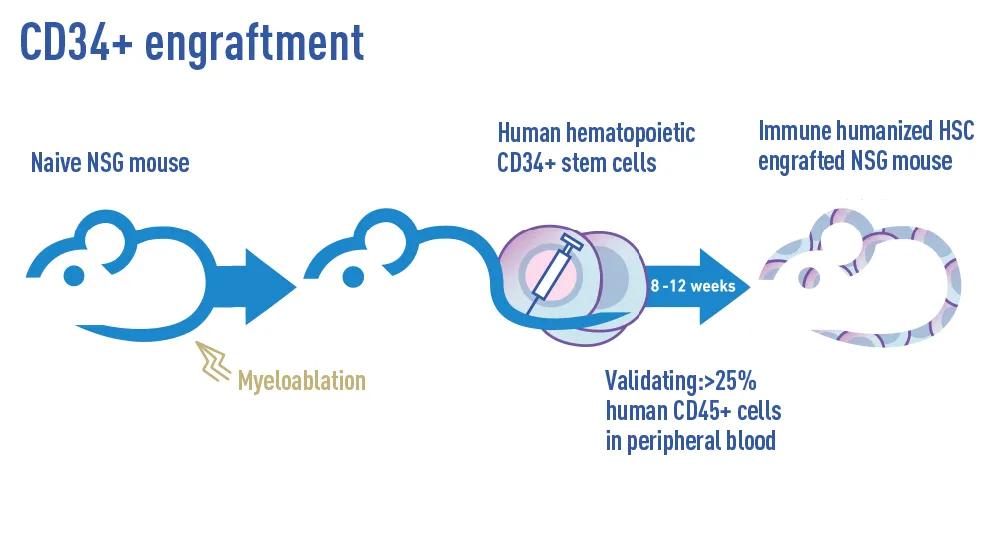
CD34+ Hematopoietic Stem Cells
HSCs are pluripotent stem cells, commonly identified by the cell surface marker CD34, and are primarily found in bone marrow, cord blood, and mobilized peripheral blood. HSCs possess two defining properties: long-term self-renewal and the capacity to differentiate into all mature blood cell types, including erythrocytes, leukocytes, and thrombocytes. As such, they form the foundation of the entire hematopoietic and immune systems. Through stepwise differentiation, HSCs give rise to all major immune cell lineages, including T cells, B cells, natural killer (NK) cells, antigen-presenting cells, and myeloid cells.
When human HSCs are engrafted into immunodeficient mice, human T cells undergo thymic selection within the mouse thymus. During this process, T cells are positively and negatively selected on mouse major histocompatibility complex (MHC) molecules, resulting in T cells that are tolerant to mouse tissues. This thymic education substantially reduces donor immune reactivity toward the host, greatly lowering the risk of graft-versus-host disease (GvHD). Some key benefits of HSC engraftment include:
Long-Term Experiments: HSC-engrafted models support longer experimental timeframes compared to PBMC-engrafted models, extending the experimental window by several months. HSC-engrafted NSG™ and NSG variants have shown persistence of human CD45 cells at 40 weeks post-engraftment (unpublished JAX data). This extended study window enables long-term in vivo investigations such as immune memory formation, rechallenge studies, gene and cell therapy evaluation, and co-engraftment of human tumors.
Diverse and Robust Immune Populations: HSC engraftment leads to the generation of multiple immune cell types, including adaptive and innate populations. These cells undergo development in vivo and exhibit functional activity, providing a physiologically relevant immune landscape. This comprehensive immune landscape is essential for studying immune cell interactions, human responses to biologics, and evaluating therapies that target multiple immune pathways.
Improved Modeling of Human Immune Ontogeny: Because immune cells arise de novo from transplanted HSCs, this engraftment approach more closely recapitulates human hematopoietic and immune development. This is particularly valuable for studying immune maturation, lineage commitment, tolerance mechanisms, and the effects of genetic or environmental perturbations on immune system formation.
Now that we know what HSCs are, let’s discuss the characteristics of JAX’s HSC engrafted mice.
JAX’s HSC Engrafted Mouse Model Characteristics
The Jackson Laboratory maintains readily available, off-the-shelf, as well as on-demand, NSG mice and NSG variants engrafted with human HSCs. Each mouse is shipped with a guaranteed minimum 25% human CD45+ leukocyte engraftment in the peripheral blood. Engraftment levels are confirmed approximately 8 to 12 weeks post-transplantation using flow cytometry. Standard panels include antibodies against human CD45+ and murine CD45+, as well as lineage-specific markers such as CD3+, CD33+, CD19+, CD56+, and others, depending on the variant engrafted. Additional markers can be incorporated upon request to meet specific experimental needs. Upon request, researchers may request specific engraftment criteria, HLA types, or donors from our inventory that best align with their study requirements.
The immunophenotypic profile of the engrafted cells varies depending on the NSG variant.1,2,3,5,7 JAX scientists have developed several unique NSG variants, each engineered to express specific human cytokines and/or growth factors that support the development and maintenance of different immune populations at defined relative frequencies. These models were developed to enable tailored immune profiles suited to different experimental applications. It is important to note that, across all variants, robust maturation of human erythrocytes or thrombocytes is not observed, and human B cells do not undergo immunoglobulin class-switch recombination under these conditions. A summary of the immune characteristics associated with each variant is provided in the table below.

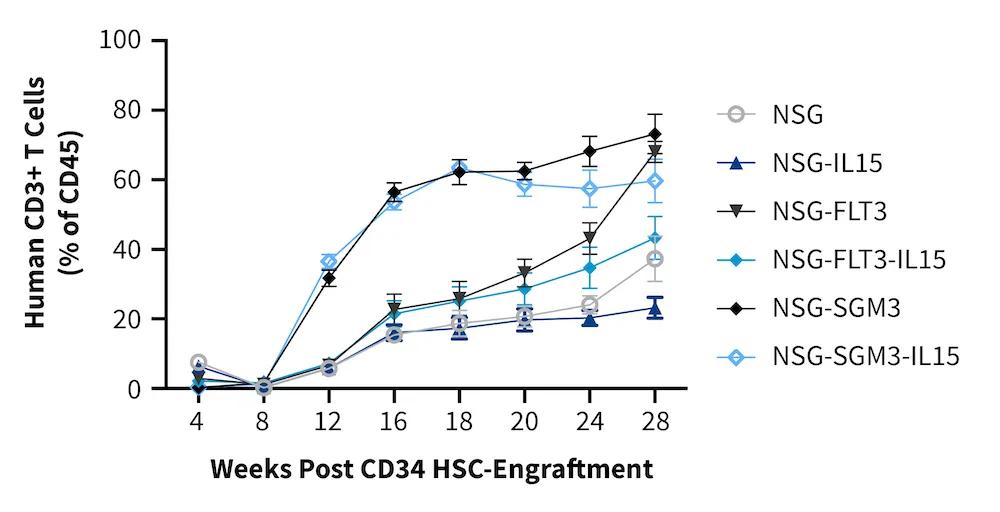
Importantly, the engraftment kinetics vary across NSG variants. To support model selection and experimental design, JAX provides extensive historical data on engraftment kinetics for multiple immune cell types in each variant. Below is a graph illustrating HSC engraftment kinetics for CD3+ T cells. Additional data on specific immune populations, in any NSG variant, is available upon request.
The reduced risk of GvHD in HSC-engrafted mice enables long-term in vivo experiments, including co-engraftment of patient- and cell-line derived xenografts (PDX and CDX). JAX offers an extensive database of low-passage solid and liquid PDX models that can be co-engrafted on any NSG variant to better recapitulate the tumor microenvironment and human immune interactions. JAX, along with others, have evaluated the engraftment kinetics of many PDXs.4,6 All JAX engraftment data is available upon request. These Onco-Hu® mice are an excellent translation model for proof-of-concept studies and for assessing the efficacy of immunotherapies.
Conclusion and Additional Resources
HSC humanized mice are an excellent platform for long-term studies where there is interest in more than one immune population and for applications involving slow-growing tumors. Explore our suite of HSC-engrafted mouse models to identify the best model for your research. Curious about all JAX's humanized mice offerings? Explore our blog, 'What is a (JAX) humanized mouse?' to learn more. Find out why drug developers are increasingly integrating immune humanized mice in their pipelines.
Don’t forget to check out our blog on PBMC engraftment to help you decide which immune humanized model is best suited for your research.
References
Aryee, K. E., Burzenski, L. M., Yao, L. C., Keck, J. G., Greiner, D. L., Shultz, L. D., & Brehm, M. A. (2022). Enhanced development of functional human NK cells in NOD-scid-IL2rgnull mice expressing human IL15. FASEB Journal: official publication of the Federation of American Societies for Experimental Biology, 36(9), e22476.
Billerbeck, E., Barry, W. T., Mu, K., Dorner, M., Rice, C. M., & Ploss, A. (2011). Development of human CD4+FoxP3+ regulatory T cells in human stem cell factor-, granulocyte-macrophage colony-stimulating factor-, and interleukin-3-expressing NOD-SCID IL2Rγ(null) humanized mice. Blood, 117(11), 3076–3086.
Brehm, M. A., Cuthbert, A., Yang, C., Miller, D. M., DiIorio, P., Laning, J., Burzenski, L., Gott, B., Foreman, O., Kavirayani, A., Herlihy, M., Rossini, A. A., Shultz, L. D., & Greiner, D. L. (2010). Parameters for establishing humanized mouse models to study human immunity: analysis of human hematopoietic stem cell engraftment in three immunodeficient strains of mice bearing the IL2rgamma(null) mutation. Clinical immunology (Orlando, Fla.), 135(1), 84–98.
Liu, J. F., Palakurthi, S., Zeng, Q., Zhou, S., Ivanova, E., Huang, W., Zervantonakis, I. K., Selfors, L. M., Shen, Y., Pritchard, C. C., Zheng, M., Adleff, V., Papp, E., Piao, H., Novak, M., Fotheringham, S., Wulf, G. M., English, J., Kirschmeier, P. T., Velculescu, V. E., … Drapkin, R. (2017). Establishment of Patient-Derived Tumor Xenograft Models of Epithelial Ovarian Cancer for Preclinical Evaluation of Novel Therapeutics. Clinical cancer research: an official journal of the American Association for Cancer Research, 23(5), 1263–1273.
Marodon, G., Desjardins, D., Mercey, L., Baillou, C., Parent, P., Manuel, M., Caux, C., Bellier, B., Pasqual, N., & Klatzmann, D. (2009). High diversity of the immune repertoire in humanized NOD.SCID.gamma c-/- mice. European Journal of Immunology, 39(8), 2136–2145.
Wunderlich, M., Chou, F. S., Link, K. A., Mizukawa, B., Perry, R. L., Carroll, M., & Mulloy, J. C. (2010). AML xenograft efficiency is significantly improved in NOD/SCID-IL2RG mice constitutively expressing human SCF, GM-CSF and IL-3. Leukemia, 24(10), 1785–1788.
Yao, L.,…Brehm, M.A. (2022). “Enhanced Development of Functional Human Innate Immune Cells in a Novel FLT3null NSG Mouse Strain Expressing Human FLT3L”, AACR.
