Immune Cell Humanized Mouse Models: PBMC Engrafted Mice
Charu Gupta, Ph.D., Senior Technical Information Scientist
Key PBMC Takeaways:
Rapid engraftment: PBMCs engraft in 2-3 days with mature, human-protein expressing T cells.
Human-relevant modeling: Better modeling of human-specific pathogenic diseases (e.g., HIV, Dengue), GvHD, human immune responses to therapeutics.
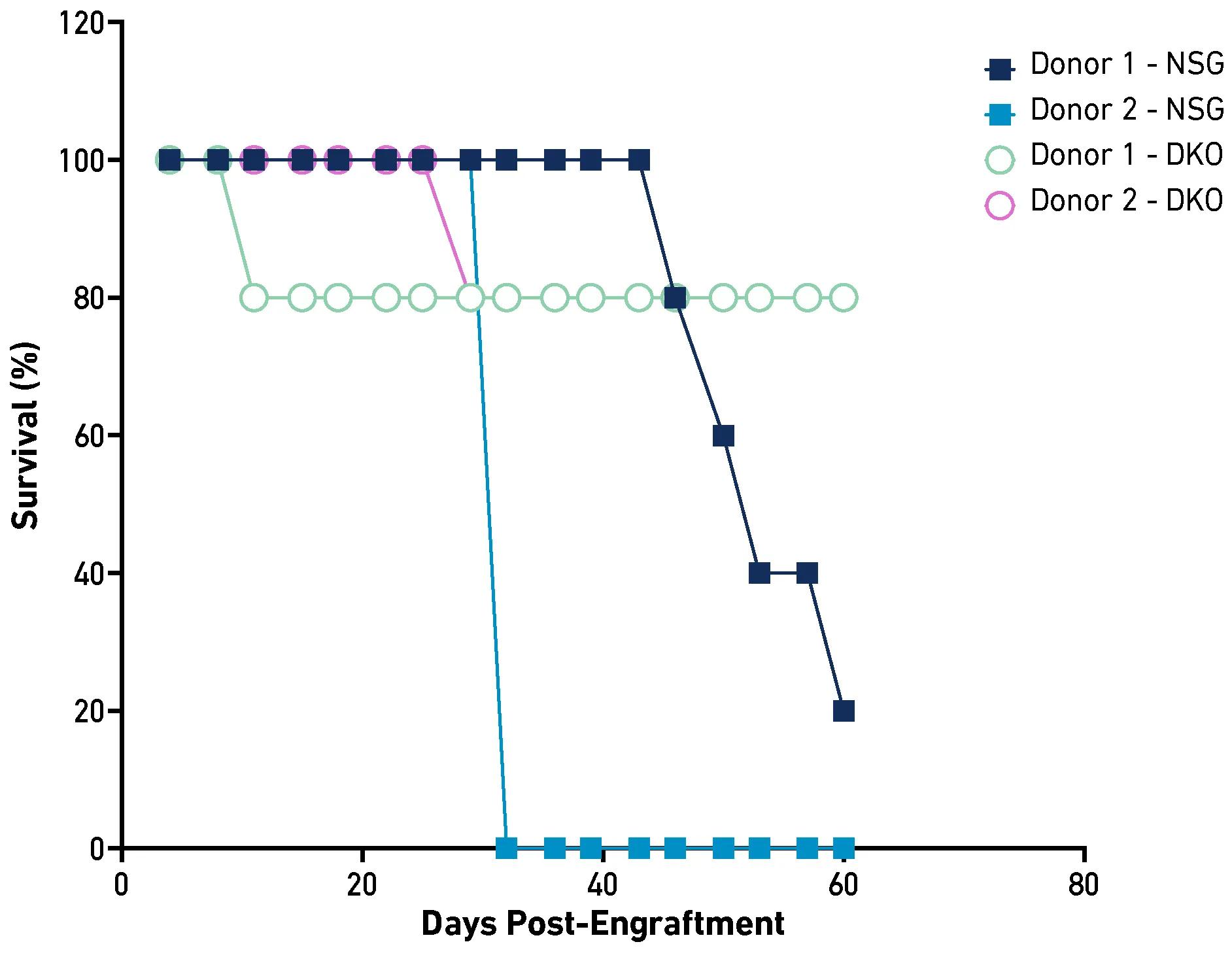
Extended study window: NSG™ DKO model delayed GvHD, enabling experiments up to 75-100 days.
Repeatable research: Pre-characterized PBMC donor engraftment kinetics and immune profile data available upon request. PBMC donor can be reserved.
An immune cell humanized mouse is a genetically modified mouse model lacking a functional murine immune system and engrafted with functional human immune cells. The human immune cells in these mice closely mimic human immune system responses in vivo, providing a valuable tool for translational preclinical research. These models enable researchers to study human-specific immune functions, disease progression, and the therapeutic potential of their preclinical drug candidates with greater accuracy.
At JAX, we specialize in two well-validated methodologies for generating immune cell humanized mice, both of which reliably recapitulate many aspects of the human immune system in a reproducible manner. These approaches are:
JAX performs both types of engraftment in multiple genetic variants of NSG™ mice and has optimized both approaches to efficiently and consistently generate humanized mice. While both engraftment approaches result in immune cell humanized mice, there are distinct differences between the two models that influence the design and outcome of experiments. Understanding the differences between these two approaches will help you choose the optimal immune humanized model for your research.
In this blog, we’ll explore what PBMCs are and the advantages they offer for your research. For a more detailed look into HSC engraftment and its experimental uses, check out our related HSC engraftment blog. These blogs aim to help you select the most suitable engraftment method for your research needs.
Peripheral Blood Mononuclear Cells
PBMCs are single-nucleus immune cells circulating in the peripheral blood. They make up a small fraction of all circulating immune cells and are composed of lymphocytes (T cells, B cells, NK cells), dendritic cells, and monocytes. T cells are the most abundant, making up approximately 60% of the total circulating PBMC population.5
Importantly, a key distinguishing feature of PBMC humanization is that the engrafted PBMC T cells are developmentally mature; they have undergone selection processing, aka “education”, in the human thymus. This selection process trains T cells to recognize human cells and tissues (i.e. self-recognition to reduce autoimmune reactions) and respond to foreign antigens presented by antigen presenting cells (i.e., non-self recognition and infectious agents) to mount an immune response.
As a result, one major benefit of PBMC humanization is that the engrafted T cells are mature and express human proteins involved in immune modulation and function. Three key benefits of this approach are:
Accurate Human Disease Modeling: PBMC humanization creates an in vivo platform that more accurately models and recapitulates human-specific pathogenic diseases, such as HIV and Dengue.2 These human-specific diseases have historically been challenging to model in conventional preclinical models, which lack the human proteins needed to replicate the disease.
Translational Modeling & Drug Testing of Immune-Related Conditions: PBMC humanization offers a more translational platform for modeling and investigating conditions involving activated immune cells (e.g., effector and memory T cells), like autoimmune disorders and cancer, as well as for testing novel T cell-targeted therapeutics.2,6,4 Since the engrafted cells are mature and functional, the immune responses more accurately mimic those in humans.
Predictive Testing of High Human-Specificity Drug Candidates: PBMC humanization offers a promising platform for testing drug candidates with high human specificity and may reduce the need for analogs in preclinical testing. In conventional models, these analogs are often necessary when drugs are inactive due to the lack of relevant human targets.3 By utilizing PBMC humanization, researchers can more effectively test and optimize their clinical compounds.
Now that we know what PBMCs are, let’s discuss the characteristics of JAX PMBC engrafted mice.
JAX’s PBMC Engrafted Mouse Model Characteristics
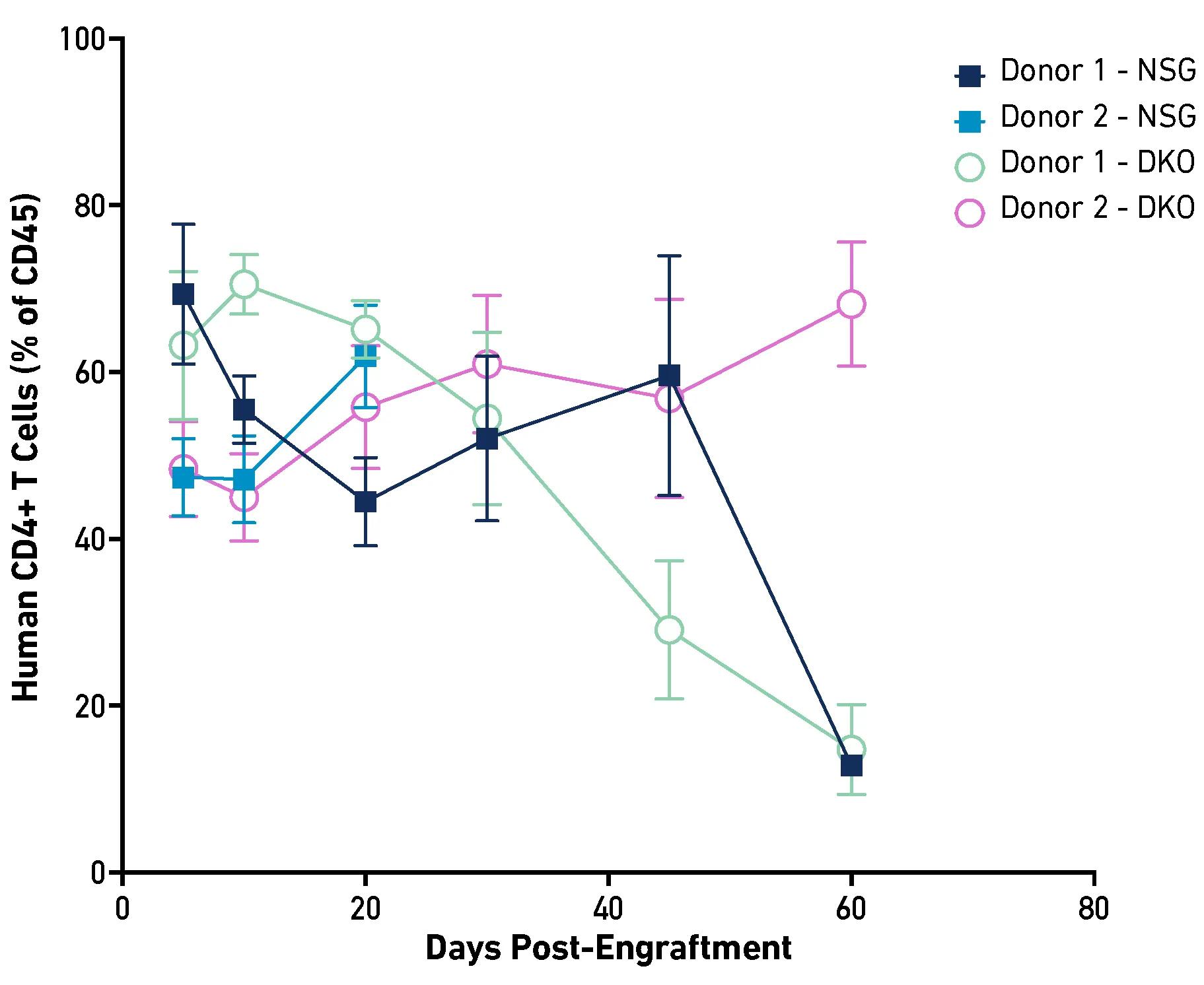
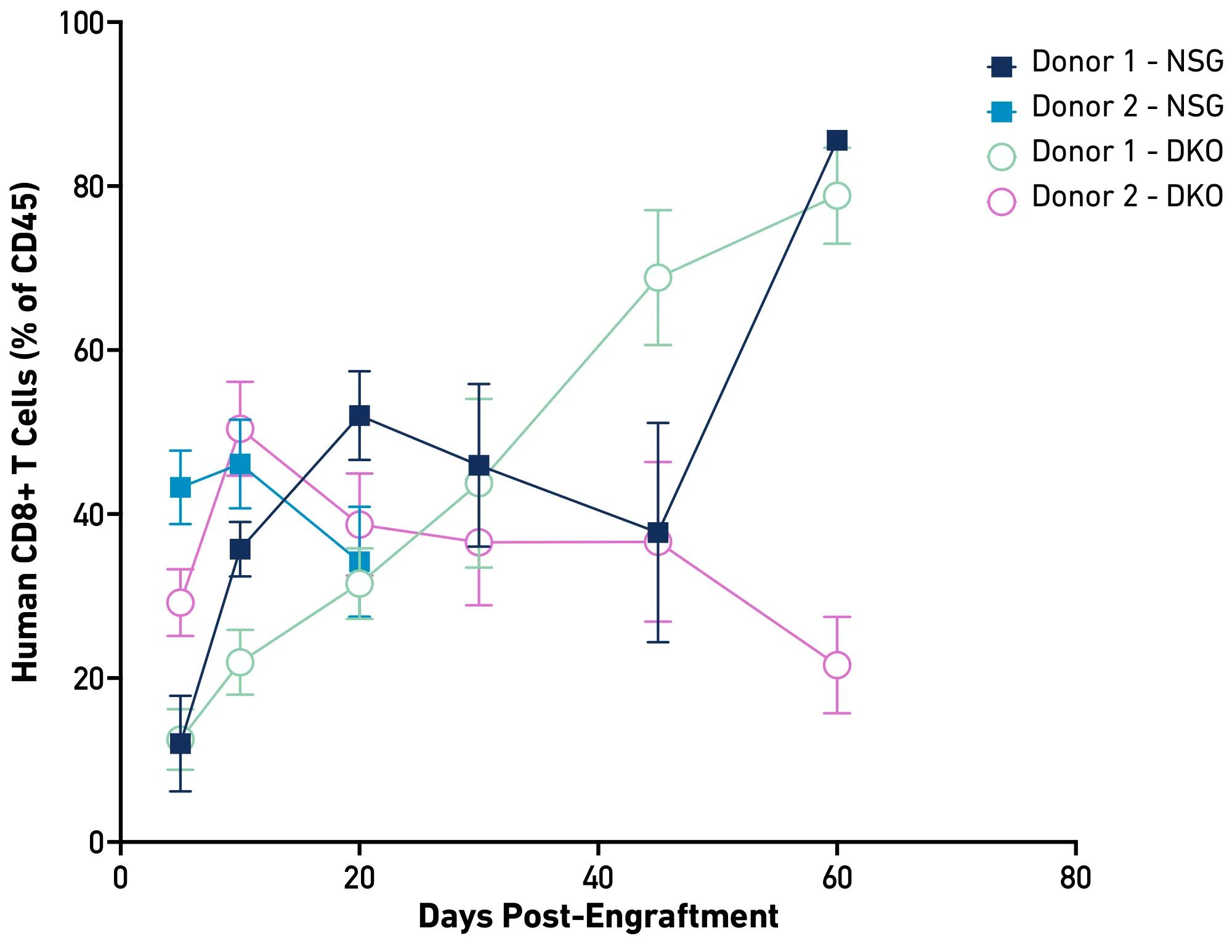
The Jackson Laboratory maintains an extensive repository of PBMCs from a diverse donor population. These resources support translational studies evaluating therapeutic potential across populations and may help identify donor-dependent variability, including the identification of potential high-risk or high-responder groups. Each donor’s PBMCs can engraft large cohorts (i.e., hundreds) of immunodeficient mice, enabling tightly controlled, statistically powered, repeatable, multi-arm in vivo experiments. Additionally, JAX offers a curated subset of pre-characterized PBMCs with defined immune cell engraftment kinetics and immunophenotypic profiles, allowing researchers to select donors tailored to specific experimental objectives. The graphs below illustrate donor-dependent differences in CD4+ and CD8+ T cell engraftment in NSG and DKO mice. Further data on donor engraftment kinetics and detailed immunophenotypic profiles are available upon request.
 |  |
At JAX, the engraftment is performed by injecting PBMCs via the tail vein into severely immunocompromised NSG™ mice or their respective strain variants. Unlike HSC engraftment, which takes several weeks to establish a human immune system, PBMC engraftment involves mature cells, leading to a rapid (2–3 days) establishment of a human immune system. The engrafted PBMCs primarily consist of T and B cells, while other mononuclear populations are generally more difficult to detect in blood greater than 10 days post-engraftment.
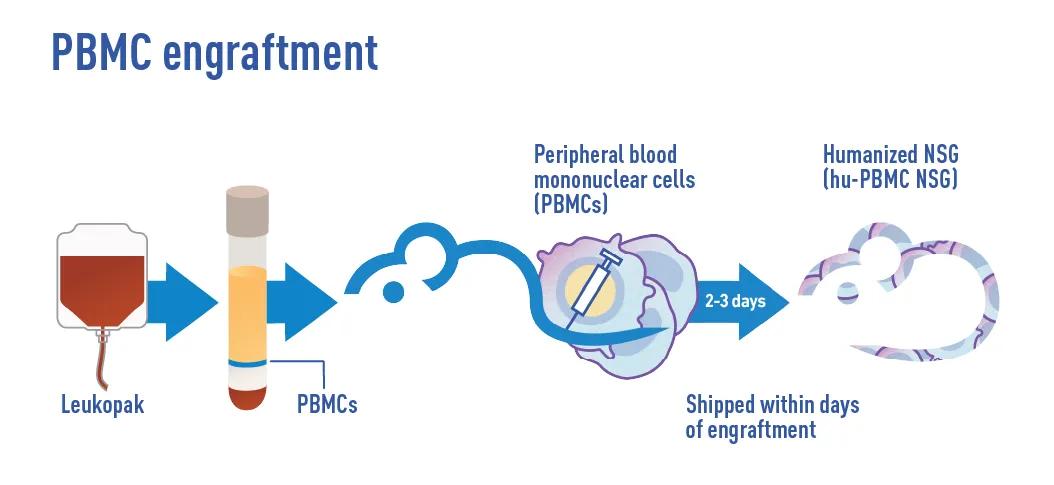
It is important to note that, because the T cells are trained in the human thymus, remaining murine immune cells and host mouse tissues will be recognized as non-self and attacked by the engrafted human immune cells. This immune response can lead to graft-versus-host disease (GvHD), a condition that can be lethal if untreated. However, this makes the PBMC model particularly useful for studying xenogeneic GvHD and evaluating the efficacy of immunosuppressive therapies. The downside is that the model has a relatively short experimental window (~2-3 weeks post-engraftment).
Recognizing the need to extend the experimental window, JAX developed an NSG MHC class I/II double knockout (DKO) mouse model (strain #: 025216). These mice lack MHC Class I and II cell surface proteins, resulting in a significantly delayed onset of GvHD post-PBMC engraftment, extending the experimental window up to 75 - 100 days post-engraftment (see graph below).1 This extension allows for the co-engraftment of patient-derived xenograft tumors and the evaluation of immune-activating therapies such as monoclonal antibodies, bi- or tri-specific antibodies as well as cellular therapies (CAR T, NK, M) for longitudinal efficacy and toxicity studies. Additionally, with the minimized GvHD risk, researchers have greater confidence that any observed toxicities result from the therapy rather than other confounding factors.2
Conclusion and Additional Resources
PBMC humanized mice have a fast engraftment rate and are an excellent platform for experiments requiring mature, activated human T cells. Explore our suite of PBMC-engrafted mouse models to identify the best model for your research. Curious about all of JAX's humanized mice offerings? Explore our blog, 'What is a (JAX) humanized mouse?' to learn more. Find out why drug developers are increasingly integrating immune humanized mice in their pipelines.
Don’t forget to check out our blog on HSC engraftment to help you decide which immune humanized model is best suited for your research.
References
Balazs AB, et al. (2011). Antibody-based protection against HIV infection by vectored immunoprophylaxis. Nature. Nov 30;481(7379):81-4. doi: 10.1038/nature10660.
Brehm MA, et al. (2019). Lack of acute xenogeneic graft- versus-host disease, but retention of T-cell function following engraftment of human peripheral blood mononuclear cells in NSG mice deficient in MHC class I and II expression. FASEB J. 33(3):3137-3151. doi: 10.1096/fj.201800636R.
Keck JG, et al. (2022). Validation of a clinically relevant humanized mouse model for the safety assessment of 4-1BB agonists utomilumab and urelumab. J Clin Oncol. doi:10.1200/JCO.2022.40.16_suppl.e14602.
King M, et al. (2008). A new Hu-PBL model for the study of human islet alloreactivity based on NOD-scid mice bearing a targeted mutation in the IL-2 receptor gamma chain gene. Clin Immunol. 126(3):303-14. doi: 10.1016/j.clim.2007.11.001.
Kleiveland CR. (2015). Peripheral Blood Mononuclear Cells. In: Verhoeckx K, Cotter P, López-Expósito I, et al., editors. The Impact of Food Bioactives on Health: in vitro and ex vivo models. Cham (CH): Springer; Chapter 15. doi: 10.1007/978-3-319-16104-4_15
Whitfield-Larry F, et al. (2011). HLA-A2-matched peripheral blood mononuclear cells from type 1 diabetic patients, but not nondiabetic donors, transfer insulitis to NOD-scid/γc(null)/HLA-A2 transgenic mice concurrent with the expansion of islet-specific CD8+ T cells. Diabetes. 60(6):1726-33. doi: 10.2337/db10-1287.